Current Events Lesson Plan December 31, 2015-January 6, 2016
Current Event: Periodic Table Scores Its Seventh Complete Row
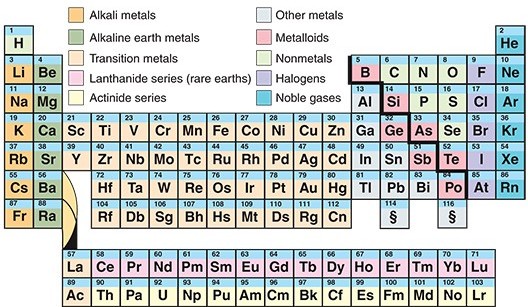
The International Union of Pure and Applied Chemistry recently recognized four new elements. The new elements, which have yet to be officially named, are numbers 113, 115, 117, and 118 on the periodic table. The discovery of these four elements fills all the remaining gaps in the periodic table’s seventh row. Each chemical element is a kind of atom. The core of an atom, called a nucleus, contains even smaller particles called protons. The more protons an element has, the heavier it is. Extremely heavy elements are unstable, tending to fall apart like towers of loose blocks. Elements 113, 115, 117, and 118 are so heavy and unstable that they do not even exist in nature. Scientists created them in the lab by smashing together the nuclei of lighter atoms. They then observed the resulting new atoms—easier said than done, because the heavy atoms disintegrate into lighter atoms and particles in just a fraction of a second.
Objective:
The periodic table is a chart that lists the known chemical elements arranged according to their characteristics. The periodic table is one of the most useful tools in science. The version of the table commonly used today organizes the elements into rows and columns. Elements in the same column share certain chemical properties. These properties repeat row by row in a regular or periodic fashion, giving the table its name. The French chemist Antoine Lavoisier first established the modern concept of the element in the late 1700’s. As more and more elements were discovered, chemists noticed that the elements show certain chemical similarities and trends in properties. The Russian chemist Dmitri Mendeleev created what scholars widely consider to be the first modern periodic table, published in 1869. The Behind the Headlines news story and related World Book articles explore other scientific topics.
Words to know:
Discussion Topics:
1. Ask your students to name some well-known chemical elements and their symbols. (Students might say Aluminum-Al, Carbon-C, Copper-Cu, Gold-Au, Helium-He, Hydrogen-H, Iron-Fe, Mercury-Hg, Nitrogen-N, Lead-Pb, Oxygen-O, Silver-Ag, Sodium-Na, Uranium-U, and Zinc-Zn.)
2. The names of some elements come from Greek or Latin words. Many elements are named for a place or an individual. Ask your students if they were to discover a new element, what would they name it?
3. Ask your students, “Which branch of science do you think is the most interesting to study—astronomy, biology, chemistry, geology, or physics?”
4. Ask your students to use World Book’s Timelines feature to view or add to the Advances in Chemistry timeline. (Students may wish to use World Book’s Chemistry article for help.)